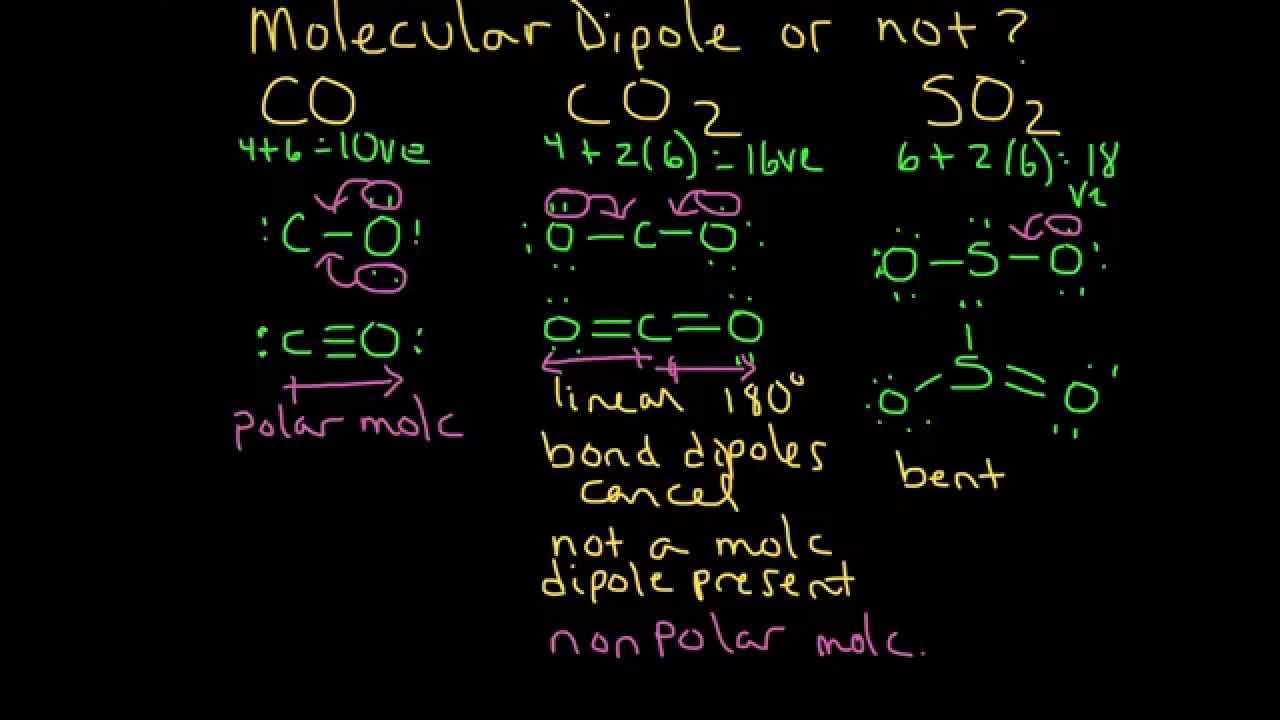Experiment shows that H2O has a dipole moment while CO2 has not. - Sarthaks eConnect | Largest Online Education Community
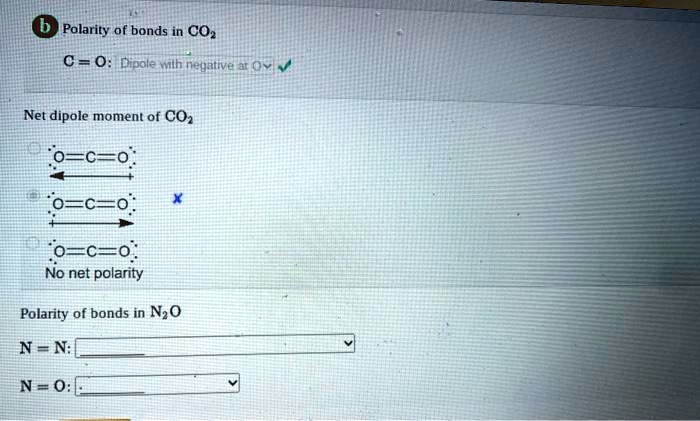
SOLVED: Polarity of bonds in CO2 C = O: Dipole Milh negative O Net dipole moment of CO2 0 = C = O; 0 = C = O; O = C =

i) Discuss the significance/applications of dipole moment.(ii) Represent diagrammatically the bond moments and the resultant dipole moment in CO_{2}, NF_{3} and CHCl_{3}.
Minerals | Free Full-Text | CO2 Dipole Moment: A Simple Model and Its Implications for CO2-Rock Interactions
Welcome to Chem Zipper.com......: Dipole moment of CCl4 is zero while that of CHCl3 is non zero. explain.
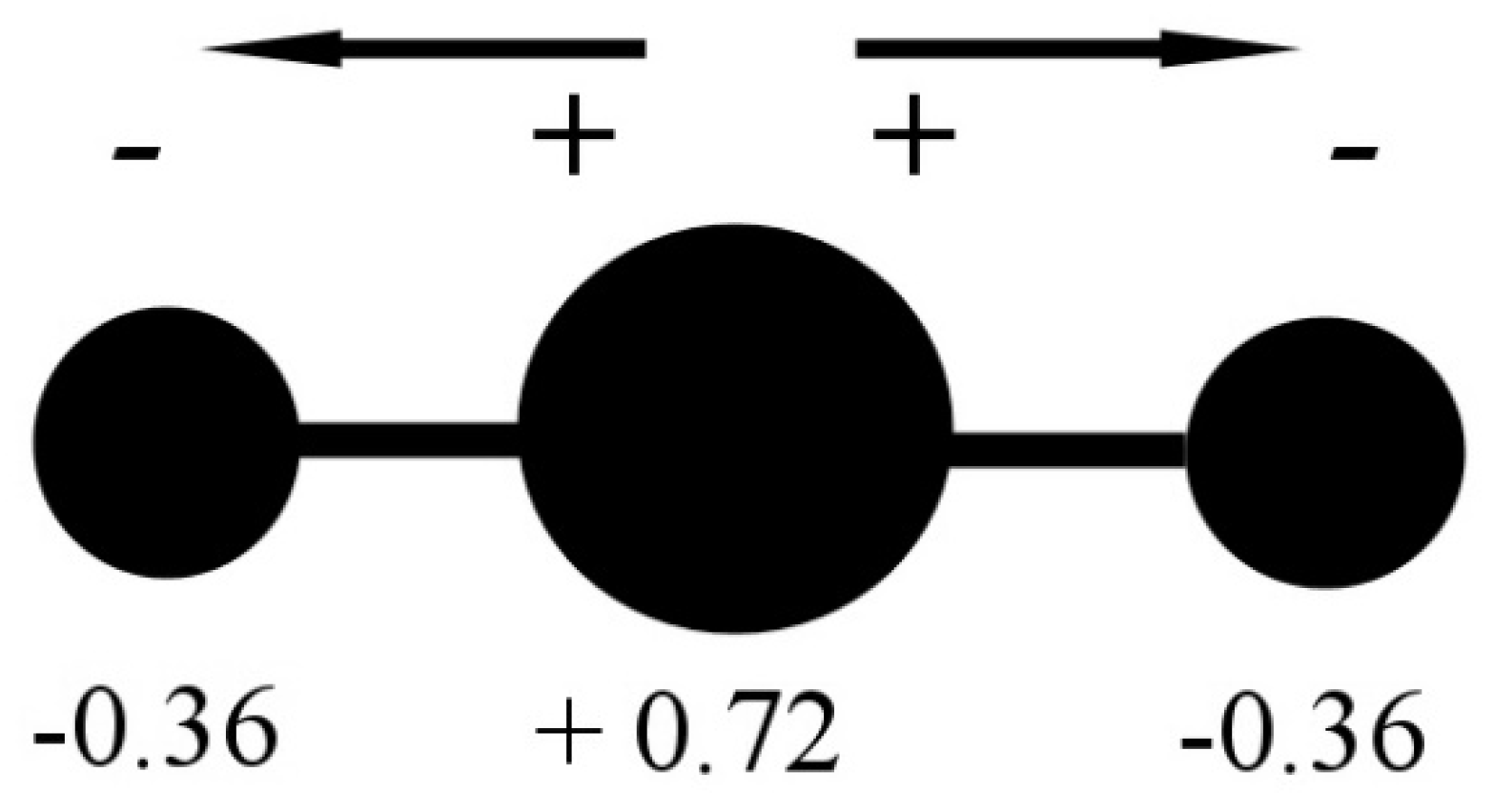
Minerals | Free Full-Text | CO2 Dipole Moment: A Simple Model and Its Implications for CO2-Rock Interactions
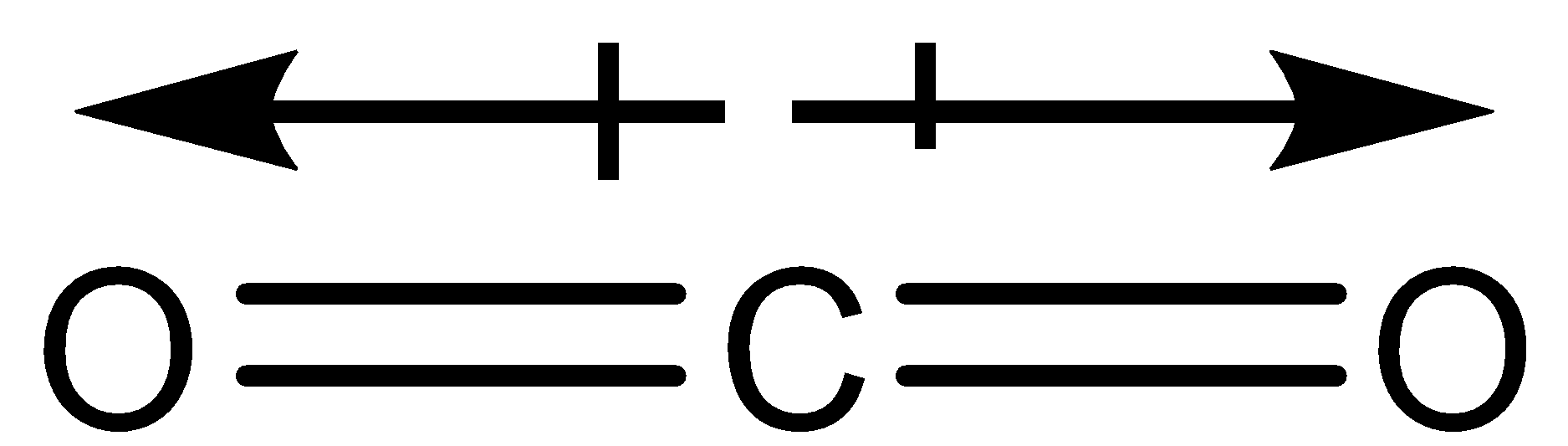
The dipole moment of $C{O_2}$ is zero, which implies that:-(A) Carbon and oxygen have equal electronegativity.(B) Carbon has no polar bond.(C) $C{O_2}$ is a linear molecule.(D) Carbon has bond moments of zero
Define dipole moment? Comment on structure & dipole moment of CO2, BF3. - Sarthaks eConnect | Largest Online Education Community
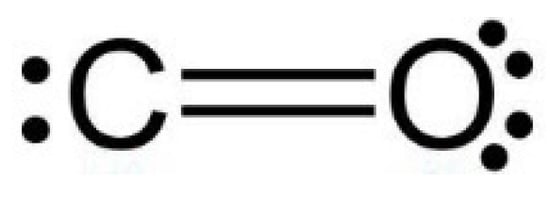
Minerals | Free Full-Text | CO2 Dipole Moment: A Simple Model and Its Implications for CO2-Rock Interactions

Determine if the depicted species has a permanent dipole moment. Carbon dioxide, CO2 | Homework.Study.com




![Punjabi] The dipole moment of CO2 is zero. Why? Punjabi] The dipole moment of CO2 is zero. Why?](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/9968028.webp)